Investigating the Core Tenet of Immunotherapy: MHC-I Peptide-Receptor Complexes
Edited by Tom Cohen
Axel Ducret is a Senior Principal Scientist at F. Hoffmann-La Roche, investigating protein and peptide quantification strategies for immunotherapeutic modalities. At Oxford Global’s Preclinical Immuno-oncology: Online conference, Ducret kicked off the symposium with a talk on using targeted mass spectrometry to investigate MHC-I peptide-receptor complexes.
The ‘How’ and ‘Why’ Of Measuring MHC-I Peptide-Receptor Complexes
Ducret introduced his talk by stating that the defining focus of modern immunotherapy is the presence of MHC-I peptide-receptor complexes that only occur on abnormal cells (e.g., cancer cells). Normally, the presence of those complexes would be enough to trigger an appropriate immune response by cytotoxic T cells. However, the problem is that this does not occur in patients whose immune surveillance is defective.
Modern immunotherapy has developed novel and innovative workarounds for this, such as CAR T therapy or bispecific antibodies. Ducret explained that “it is essential that the researchers ensure the specificity of these therapies.” In particular, this means making sure that the mode of action is dependent on the presence of the target antigen and killing efficiency is proportionate to the antigen density.
Ducret described the concept of MHC-I associated peptide proteomics (MHC-I MAPPs), to tackle this difficult problem. They start with cell lines and tissue samples that they lyse and then perform immune enrichment. The addition of internal standards (heavy labelled MHC-I peptides) ensures that the ensuing analysis using targeted mass spectrometry reports quantitative data.
Example 1 – Targeted Protein Degradation
Ducret characterised this technique using two examples. The first was detecting and quantifying BRD4-derived MHC-I peptides that had been generated using PROTACs. The concept is to induce proteasomal protein degradation by recruiting E3 ligase to the target protein.
- PROTACs: “Exciting Opportunities to Explore New Biology”
- Getting PROTACs into the Clinic: The Challenges
“Interestingly,” said Ducret, “targeted protein degradation using PROTACs is achieved by the exact same pathway that generates MHC-I peptides.” Ubiquitinated proteins are degraded into peptide fragments, which are then loaded onto MHC-I receptors to be presented on the cell surface.
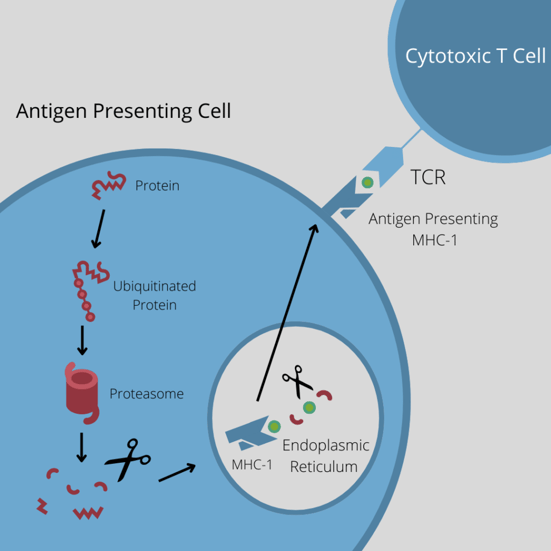
According to Ducret, the strategy of using PROTACs is an interesting way to investigate whether it is possible to change the composition of the MHC-I peptidome.
The team at Roche looked specifically at the BRD4 degraders as inducers of peptide presentation. Ducret explained that the protein BRD4 is critically involved in the malignant disease maintenance of acute myeloid lymphoma (AML) and degraders of BRD4 have been shown to be pre-clinically efficacious.
“We asked ourselves: could part of the mode of action of these degraders be the immune system itself, via the enhanced presentation of BRD4 peptides?”
“So, we asked ourselves: could part of the mode of action of these degraders be the immune system itself, via the enhanced presentation of BRD4 peptides?” Ducret explained. To prove this, Ducret and his team designed an experiment to see whether BRD4 degraders increased the abundance of endogenous BRD4 peptides bound to the MHC-I receptors on the cell surface. Furthermore, if this were the case, it should also be shown that it is “dose dependent, time dependent, degrader dependent, and in multiple cell lines.”
The team started their experiment using a proof-of-concept. They used a molecule that was described to be active in preclinical models, called dBET1. In this molecule, one arm binds to the E3 ligase, the other binds to the target, BDR4. From this, they managed to demonstrate that BRD4 was very specifically degraded using the treatment.
As the experiment was successful, they looked into the MHC-I peptidome and detected five BRD4-derived peptides, selecting two of them for quantification: AEALEKLFL (‘AEA’) and QEFGADVRL (‘QEF’). It was shown that the levels of the two peptides presenting at the MHC-I receptors increased over the concentration of dBET1.
In the second experiment, Ducret’s researchers wanted to demonstrate that the effect was actually the effect of the active degraders rather than anything else. They compared the effect of dBET1 to dBET1-(R), which is an inactive isomer of the active molecule and has no binding to BRD4, and to dBET1-NMe, which has a methyl group preventing its binding to CRBN. They also tested the two active moieties separately, JQ1, which binds to BRD4, thalidomide which binds to CRBN, to demonstrate that BRD4 degradation was only occurring using the full active PROTAC molecule.
The results were clear: The only conditions for which they had BRD4 degradation were when using the active dBET1 molecule. This was shown even more clearly using the peptidomic approach, where the only conditions that showed an increased presentation of the two peptides were again using the active PROTAC molecule.
Example 2 – Bispecific Antibodies
The second example that Ducret showed us was using MHC-I MAPPs to support the development of a novel MHC-I peptide-specific T cell bispecific antibody. A bispecific antibody’s job is to recruit cytotoxic T cells to the cancer site; an effective way to treat cancer.
“…we knew we were on a very difficult case.”
Roche was planning to enter a phase I clinical trial with a bispecific antibody that targets a MHC-I peptide-HLA receptor complex derived from the Wilms tumour protein (WT1), which is overexpressed in leukaemia as well as several solid cancer indications. “However, when were contacted by the preclinical team to work on this particular molecule, we knew we were on a very difficult case,” said Ducret.
As Ducret acknowledged, “whilst the target has been predicted to exist and was successfully targeted in other clinical trials, this particular peptide-receptor complex has never been previously characterised by conventional methods.” The preclinical team asked Ducret’s lab to develop a specific mass spectrometry detection for quantification of the peptide-receptor complex. “This was to support the mode of action and to ensure that it was a specific and safe bispecific antibody.”
First, Ducret’s team built an assay and defined the assay conditions because the RMF peptide was known to rapidly oxidise. Next, they established Limit of Detection and Lower Limit of Quantitation and they established the linearity of the assay in cellular models displaying the peptide-receptor complexes. Ducret’s team were reassured by the fact that their experiments showed the assay working in a linear manner and was sensitive enough to detect the peptide.
After these successful experiments, they worked on confirming their results using a cell line that would express the complex endogenously. They selected the SKM1 cell line because they knew that it overexpressed the WT1 protein and because it is highly sensitive to T cell-mediated killing when treated with the WT1-TCB reagent; the logical conclusion was that the cell line would present the RMF peptide. They managed to confirm the presence of the RMF-MHC-I receptor mediated complex – but only at a very low level. Still, it did demonstrate that the complex existed in endogenous levels.
Ducret’s team then went on to investigate whether they could see the complex in blast cells from HLA-A*02 positive patients with acute myeloid leukaemia. They used a WT1RMF-pMHC tool antibody, which has the same specificity as the clinical compound. From this they were able to detect a very high level of HLA-A*02 receptor complex.
Conclusions
Ducret determined that their experiments had showcased the successful development and application of targeted mass spectrometric-based workflows for the measurement and relative quantification of enriched MHC-I receptor complexes.
They had demonstrated that the use of degraders increased the level of ontogeneous MHC-I peptides for BRD4. Furthermore, they showed that they were able to identify and quantify RMF-HLA-A*02 receptor complex, which supports the mode of action for Roche’s new WT1-T cell bispecific antibody.
“It is clear that the implementation of MHC-I MAPPs was important because it demonstrates receptor complex specificity and you can precisely quantify the MHC-I-peptide-receptor complex,” concluded Ducret.
Our upcoming Immuno UK: In Person conference will be this year’s best way to gain valuable insights into the approaches impacting the immunotherapy field. Through 40+ outstanding presentations tackling key discussion points in immuno-oncology, immunology, and inflammation.