When to Cut and When to Inhibit? A Low-Throughput, Hands-On Investigation Of CRISPR-Cut and CRISPRi Efficiencies In iPSC And Neural Cells

Presented by: Pia Johansson, Scientific Coordinator at Lund University
Edited by: Ben Norris
Pia Johansson, Scientific Coordinator of the Cell and Gene Therapy Core at Lund University, spoke at Oxford Global's Gene Therapy Development &Manufacturing 2022 conference about the applications and efficiencies of different CRISPR gene editing techniques in neural cells.
CRISPRi Versus CRISPR-Cut
“Today, I'm going to talk about when to use CRISPRi and when to use CRISPR-cut,” opened Johansson. Her presentation focused on the different applications of genomic editing technologies to affect gene knockdown in target cells. Gene knockdown can be achieved by manually reducing the expression of a particular gene, whereas gene knockout sees the genes expression stopped altogether. Knockdown of genes can be obtained either through genetic manipulation, through CRISPR, or with reagents such as RNA oligonucleotides or short DNA strands.
- Key Industry Insights in Gene Therapy Manufacturing
- Refining Analytics for Gene Delivery Systems
- Using 3D Cell Culture Models in Your Research
The operative agents in CRISPR are the many varieties of Cas proteins found in bacteria, where they assist in defending against viruses. Of these, the Cas9 protein is the most widely-used. “CRISPR knockout is the normal Cas9, where the single-guide RNA guides the Cas9 to a specific sequence in the genome and then it cuts it to create a double-stranded break,” said Johansson. “Then we have CRISPRi, which uses the same technology apart from the fact that the Cas9 now has mutated sequences, which means that it no longer cuts the DNA but just binds there. Then you can fuse a KREB domain to it, which inhibits the expression of nearby genes.”
When Johansson joined the group of Molecular Neurogenetics at Lund University, work was already underway using CRISPR-cut in undifferentiated human embryonic stem cells. “The knockout efficiency was extremely high,” she said. Johansson's remit was to focus on a new cell type with new genes: in this case, the focus was on using forebrain-centric neural progenitor cells, which are more specifically differentiated. The aim of her work was to add a lentiviral vector in the middle of differentiation, with the goal of seeing what the outcome was of knocking out target genes during development.
Gene Knockout Versus Gene Knockdown
Johansson incorporated two different strategies for knocking out target genes. The first involved an all-in-one lentiviral construct, where both the guide RNA and the Cas9 were in the same vector together with green fluorescent protein (GFP). “Then, we ran a similar one where we only had the guide RNA virus and the Cas9 was constitutively expressed in the cell,” Johansson continued. However, a very high editing efficiency is required for this to be successful, as the process must work in bulk.
“Efficiency is dependent on both dose and the guide that you're using.”
“When we added the lentiviral vectors, we found very low editing efficiency and a lot of problems with survival because the sample was no longer growing at the correct density because of the vector mediated toxicity,” added Johansson. Following this, a switch was made from using CRISPR-Cut to using CRISPRi genetic perturbation. Subsequent analyses found that CRISPRi efficiency is both guide- and dose-dependent. “The first thing we found is that the efficiency is dependent on both dose and the guide that you're using.”
The first knockdown was seen in IPS material, with very high knock-down efficiency, “With a CRISPR knockout, this is obviously so much better,” said Johansson. “Because we want to use them in bulk, I think a 95% success rate is very good.” Johansson observed that the vector was very long-lasting, with CRISPRi efficiency both stable and long-term. “In the process of achieving this even the histone H3K4me3 activation mark was lost, making the overall process comparable to gene knockout.” She added that gene knockdown was still visible two and four months into the cerebral organoid differentiation, showing that the vector is very long-lasting.
Lentiviral Vectors and Gene Multiplexing
Johansson then showed an experiment where they added another lentiviral vector to see if they could knock down two genes, efficiently, at the same time. She mentioned that she was concerned whether there would have been enough dead Cas9 present for this to succeed. “But we actually have very nice knockdown of both of the genes,” she continued. This is difficult to execute if the aim is traditional knockout as this then leads to multiple double stranded breaks, which can create additional complexities within the genome.
The benefits of CRISPRi compared to CRISPR knockout include an absence of DNA breaks, as well as an easier approach to multiplexing, and it has a much faster readout. “If you don't want a complete knockdown, you can add a bit less of the viral vector, and then you will have some expression left,” said Johansson.
Johansson added that a detriment was the involvement of lentiviral vectors. “You have to be able to work with lentiviral vectors,” she explained, “and you have to be able to have someone produce them, there's cloning involved, and your cells have to be able to tolerate them.”
Future Challenges and Opportunities for Gene Editing
Rounding off her presentation, Johansson said that the benefits of performing CRISPR analysis in bulk are quite extensive, with other parties sometimes too focused on taking a clonal approach. “The population has much more genetic stability than an individual clone,” she said, “as genetic instability in clonally expanded cells is quite common.” However, when using the cells in bulk the quantity of cells involved cancels this out.
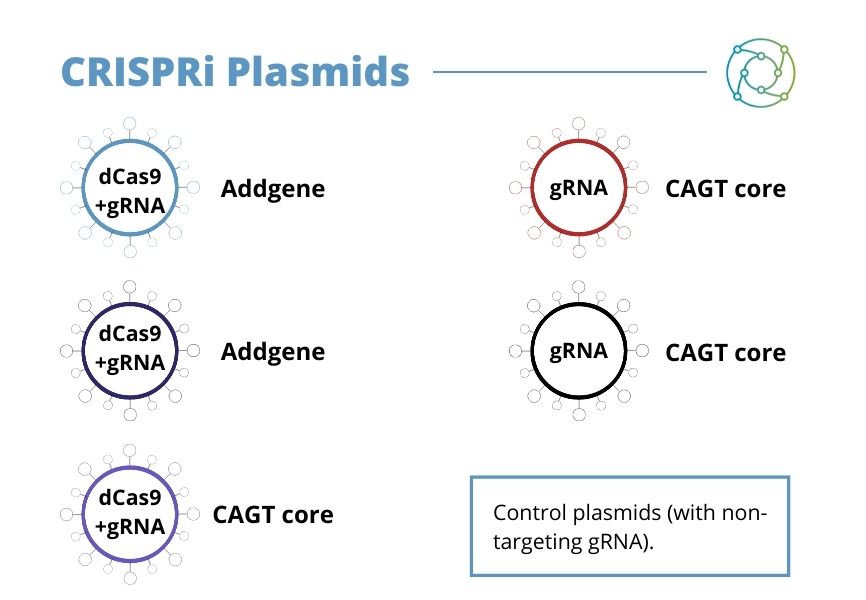
Johansson concluded by reiterating some of the different CRISPR plasmids which have been used in the process and some of which are available from the CAGT core.
Visit our Cell portal to read more articles on gene editing and the latest advances in cellular therapies. If you'd like to register your interest for our upcoming Cell UK: In-Person event, click here.







